The immune system has to fight off hundreds of microbial invaders every day, such as the human-pathogenic fungus Aspergillus fumigatus. The fungal conidia can reach the lower respiratory tract, swell and form hyphae within six hours causing life-threatening invasive aspergillosis. Invading pathogens are continuously recognized and eliminated by alveolar macrophages (AM). Their number plays an essential role, but remains controversial with measurements varying by a factor greater than ten for the human lung. We here investigate the impact of the AM number on the clearance of A. fumigatus conidia in humans and mice using analytical and numerical modeling approaches. A three-dimensional to-scale hybrid agent-based model (hABM) of the human and murine alveolus allowed us to simulate millions of virtual infection scenarios, and to gain quantitative insights into the infection dynamics for varying AM numbers and infection doses. Since hABM simulations are computationally expensive, we derived and trained an analytical surrogate infection model on the large dataset of numerical simulations. This enables reducing the number of hABM simulations while still providing (i) accurate and immediate predictions on infection progression, (ii) quantitative hypotheses on the infection dynamics under healthy and immunocompromised conditions, and (iii) optimal AM numbers for combating A. fumigatus infections in humans and mice.
Aspergillus fumigatus – macrophage interaction
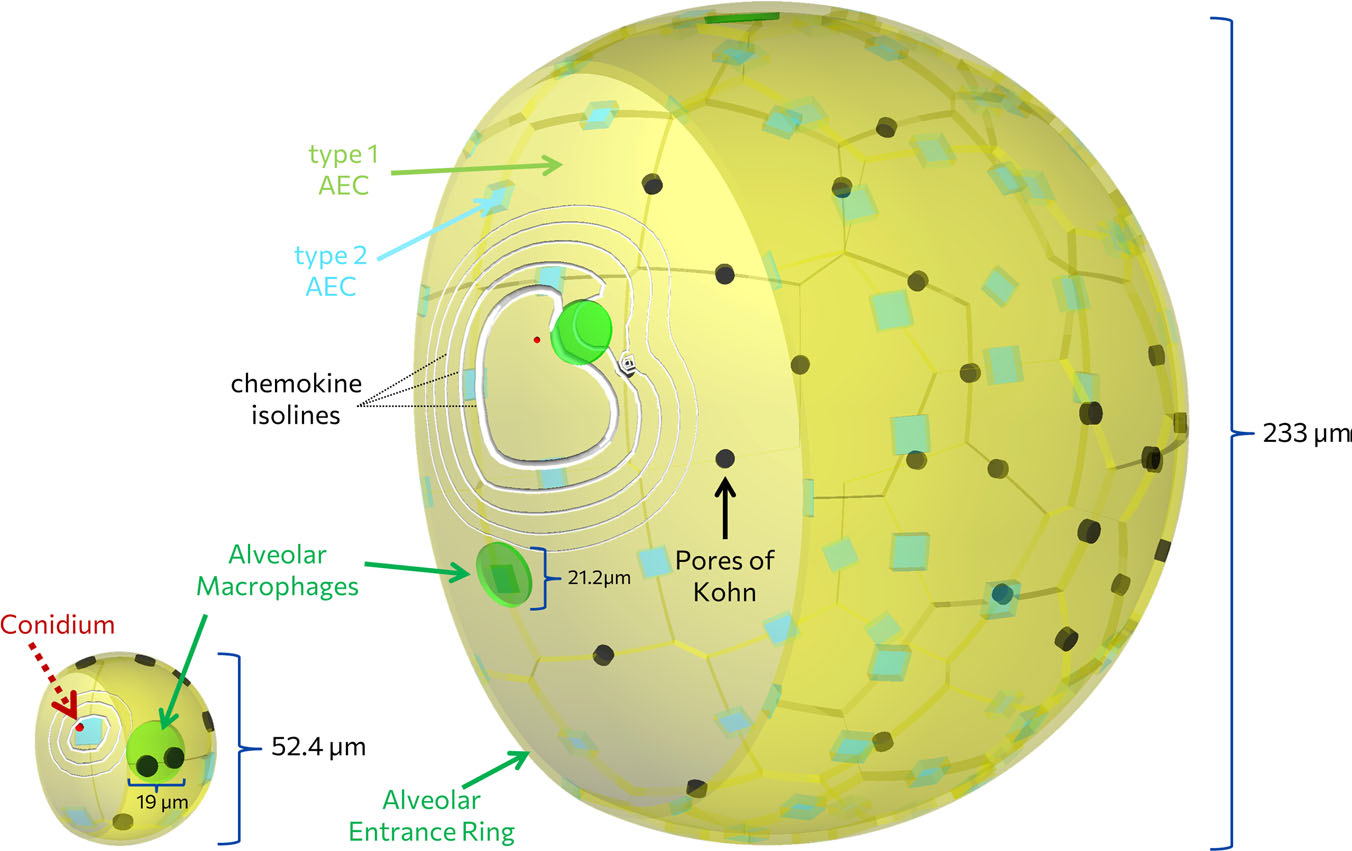
The first line of defence against the airborne human-pathogenic fungus A. fumigatus are the resident immune cells in the human…
Aspergillus fumigatus infection in virtual alveolus
Lung pathogen A. fumigatus forming hyphae and macrophages eliminating the fungus, influencing infection dynamics, studied through a hybrid agent-based model.…